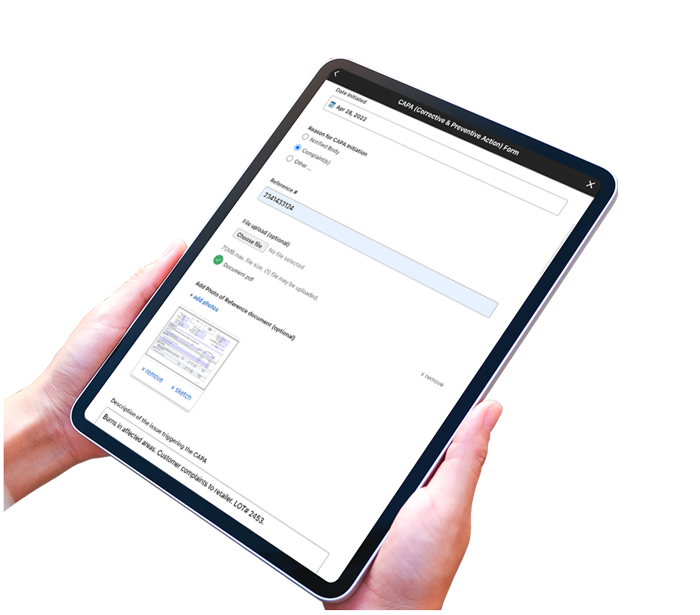
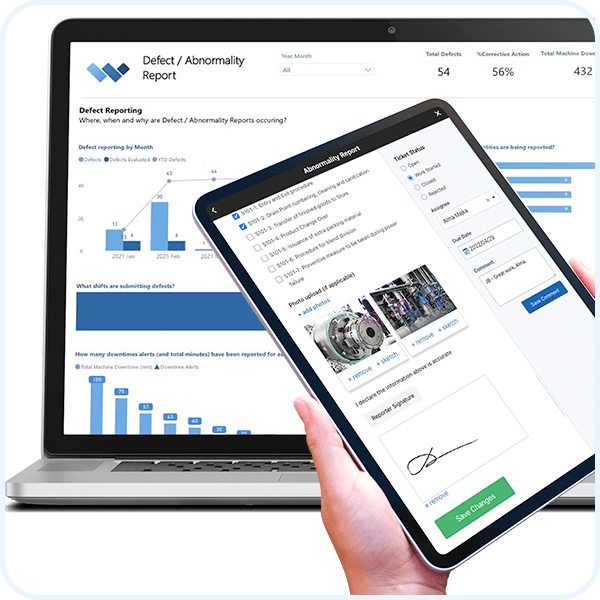
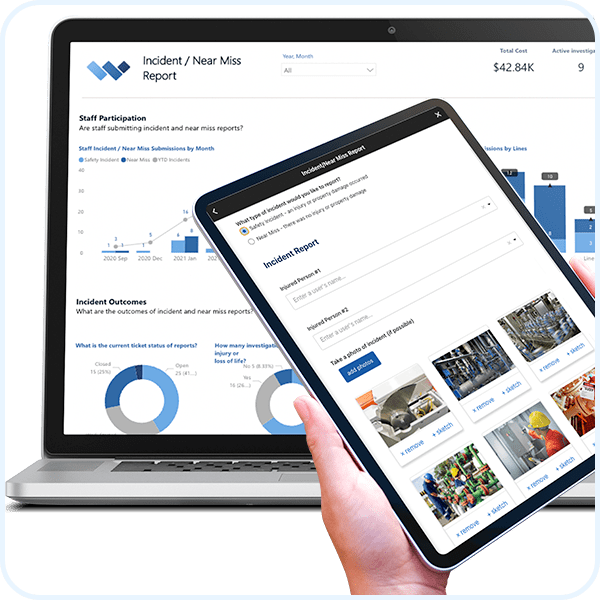
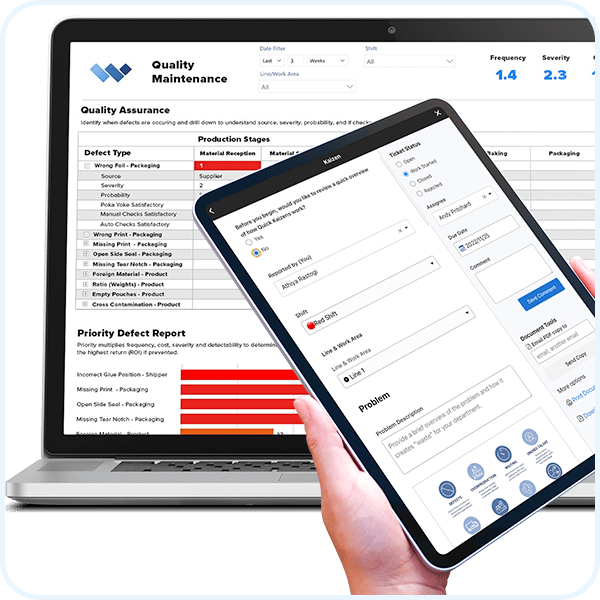
Corrective & Preventive Actions (CAPA)
OVER COMPLETED WORLDWIDE.







Marks

Diageo

Niagara Bottling

Walmart

PepsiCo logo

McDonald's

Unilever

Monin

Hello Fresh

Rise Baking

Rockwool

Canadian Tire

SportChek

Greyston Bakery

Bell

Husqvarna

Home Hardware

Automate administrative tasks, simplify work and unlock the full potential of your frontline employees.
We work with organizations of all shapes and sizes who are frustrated with paper forms and spreadsheets. They want to save money, become more efficient, and spend their time more wisely. If that sounds like you, you've come to the right place.
Everything you need to get it done.
Customizable Templates
Dashboard Reporting
Automated Workflows
Schedule Management
Real-time Visibility
On-Demand Instructions
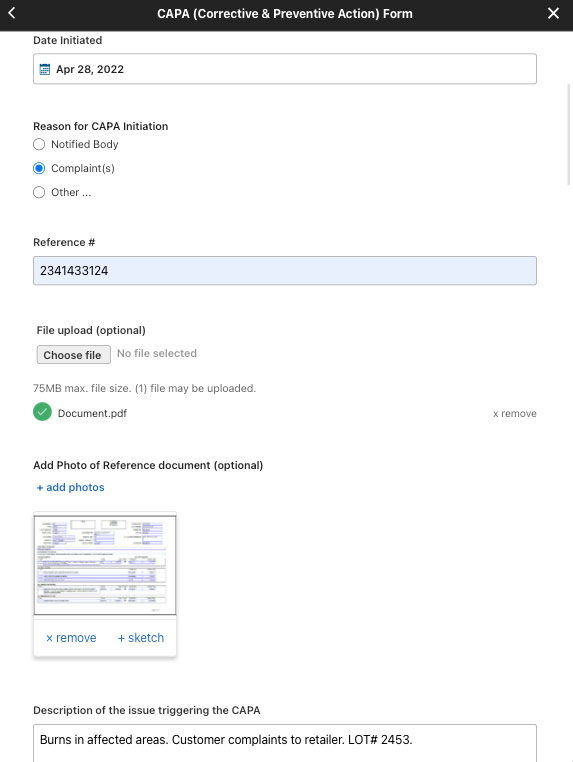

Digital DATA CAPTURE
Get it done correctly every time.


WORKFLOW AUTOMATION
Evaluate and approve.

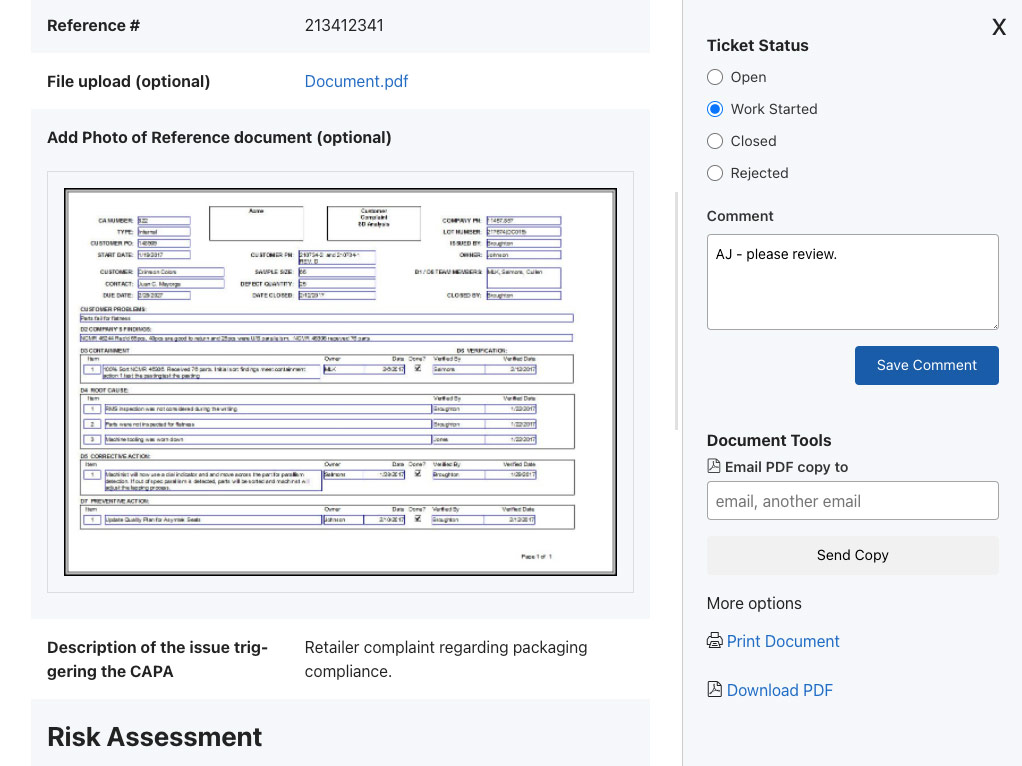

WORKFLOW AUTOMATION
Evaluate and approve.
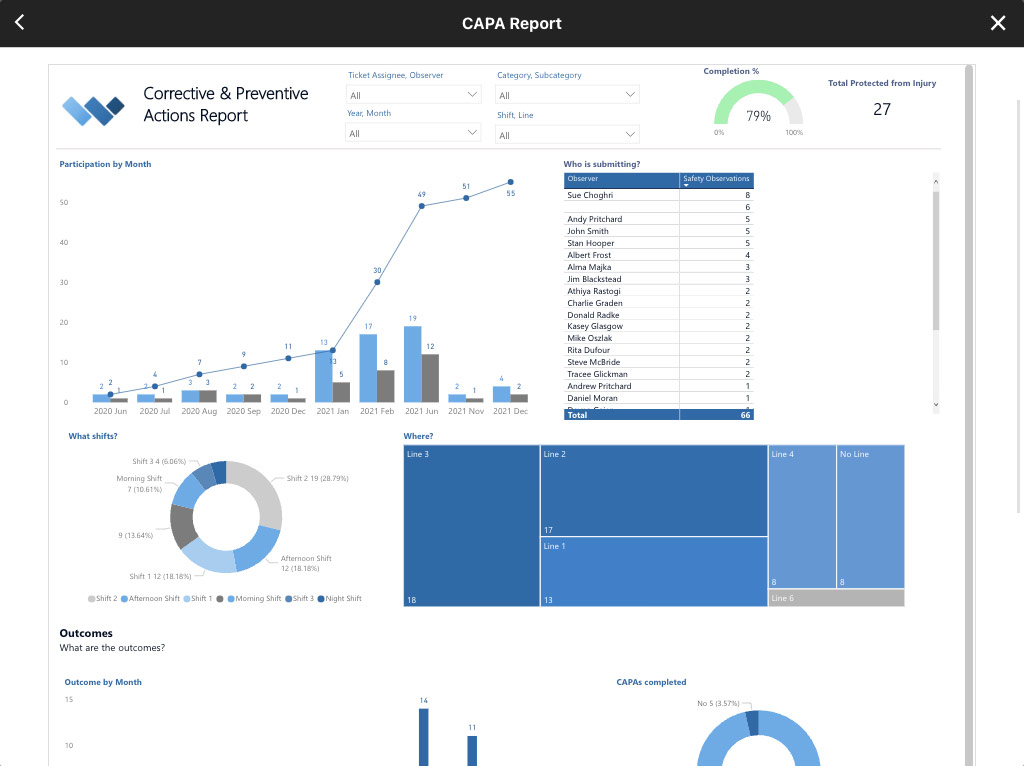
REAL-TIME REPORTING
Automate Business Intelligence.

100K+
Global Users
MILLIONS
of jobs completed
11
Languages
"The Weever suite is easy to understand and the support from the team has been amazing!"
Laura Curtis - Operational System and Process Manager, HelloFresh
"Weever saves me time, makes data visible and drives results!"
DENAIR M. - Training Manager
"Weever has changed how I run our business."
Ingrid K. - Plant Director
"Weever is extremely easy to use and simple to manage."
NICKI V - CI Engineer
"Weever gives me immediate insights into my business."
COLIN H. - Operational Excellence Manager
"The Weever team are extremely helpful and are always on hand to help with any questions or queries we may have."
Mel Cadle - Op Ex Lead Process Engineer, HelloFresh




Learn about the Platform.
Download the brochure to get all the details about how Weever works.
Do it all with Weever.
Weever is a comprehensive workflow management platform that is used to automate safety, quality, maintenance, production, compliance, continuous improvement and training operational requirements.
SCHEDULE A DEMO
Take a Guided Tour.
In just 30 minutes you will learn about customizing and using forms, workflow automation, reviewing reports, and sharing data with other business systems.
In just 30 minutes you will learn about customizing and using forms, workflow automation, reviewing reports, and sharing data with other business systems.